Malaria threat and gene therapy
At the annual meeting of the American Society of Tropical Medicine and Hygiene (ASTMH) this week, researchers presented data that indicate that the world’s most common malaria parasite is becoming a bigger threat. MedZine tells you what is happening. In addition, MedZine also highlights another study, showing successful persistent gene therapy without suppressing the immune system.
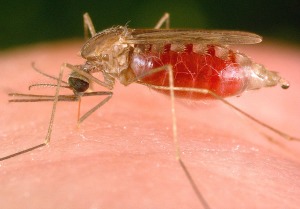
Evolving malaria is a threat
The Plasmodium vivax parasite is the most common cause of malaria and was responsible for nearly 20 million cases in 2010. In large parts of Africa, the majority of the population has been considered protected from vivax malaria, because they lack the Duffy blood group protein on their red blood cells. The absence of this protein prevents the entry of vivax malaria parasites into red blood cells. However, over the last five years there has been a growing number of reports from Africa and South America of infections in people who are Duffy-negative and should be resistant to vivax malaria.
Zimmerman and colleagues studied the genome of Plasmodium vivax types in different part of the world. The results will be published in two articles on November 21th and December 5th in PLOS Neglected Tropical Diseases. The researchers found that parasites gathered in Madagascar, where many Duffy-negative infections occur, often have two copies of the gene that encodes the parasite's Duffy-binding protein, while this was not found in parasites from regions that do not have a population consisting of both Duffy-negative and –positive individuals. In a second study, the researchers found a previously unknown gene that could be an invasion protein for gaining access to red blood cells in parasites from Cambodia. This new gene was found be to widely present in contemporary vivax parasites around the world, except for the vivax parasite used in 2008 to sequence the Plasmodium vivax genome.
Gene therapy without immune suppression
The key to successful gene therapy is the effective delivery and maintained expression of healthy copies of a gene into tissues of individuals with a disease-associated genetic mutation. In clinical trials, recombinant adeno-associated virus (rAAV) vectors have shown promise as effective therapies for several genetic diseases, including Parkinson disease and hemophilia. However, this often results in development of an immune response against the viral capsid and therefore immune suppression is frequently needed. In a publication in the Journal of Clinical Investigation, Mueller and colleagues describe a strategy that provides persistent gene therapy without the need of immune suppression. The researcher induced rAAV-mediated expression of the gene encoding M-type α-1 antitrypsin (M-AAT) in patients that were AAT deficient. Multiple intramuscular doses without immunosuppression were given to the patients. The researchers describe sustained M-ATT expression in muscle tissue for at least one year, despite an initial influx of immune cells.
Sources: Eurekalert, ASTMH, and Journal of Clinical Investigation