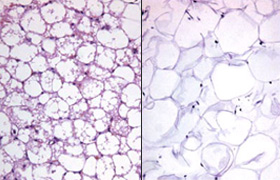
White fat turns brown when exposed to epinephrine
Cold-activated brown fat cells burn fat to produce warmth, instead of storing it like white fat cells. However, according to scientists from the US and Greece, it is now possible to turn white fat cells into brown fat cells in humans. This discovery opens up new opportunities for the fight against obesity, the research groups write in Cell Metabolism.
Brown fat cells are characterised by a high number of mitochondria and expression of the protein UCP1, which elevates the oxidation of fatty acids and glucose and sensitises cells to insulin. Previous research has revealed that UCP1 is activated by epinephrine, for example during an adrenergic response of the body. This can be triggered by cold, but also by burn injuries, which elevate epinephrine levels lasting up to several weeks.
Therefore, the researchers collected white fat cells from 48 patients (including 42 children) with serious burn injuries at several time points during their hospital stay. To analyse the brown fat characteristics of white fat cells, they determined the number of mitochondria per cell and measured UCP1 expression. This data clearly showed a partial transition of white fat to brown fat, increasing with epinephrine exposure time. Furthermore, the effect was not only apparent in children, who naturally have a high brown fat percentage, but also in adults.
Notably, the metabolism levels of white fat cells were not nearly as high as those of brown fat cells with a twenty-fold difference. However, this is favourable for future therapy, the scientists argue, since an absolute increase of energy expenditure in white fat cells would create a too high daily calorie demand.
After demonstrating the feasibility of transforming white human fat cells in vivo, the researchers are now investigating the underlying mechanism. Moreover, they will have to find a safe way to induce the adrenergic response seen in burn victims in people without burn injuries.
Sources: Eurekalert, Cell Metabolism
MedZine writes about notable science twice a week.